 W
WA gel is a semi-solid that can have properties ranging from soft and weak to hard and tough. Gels are defined as a substantially dilute cross-linked system, which exhibits no flow when in the steady-state, although the liquid phase may still diffuse through this system. A gel has been defined phenomenologically as a soft, solid or solid-like material consisting of two or more components, one of which is a liquid, present in substantial quantity.
 W
WAgar, or agar-agar, is a jelly-like substance consisting of polysaccharides obtained from the cell walls of some species of red algae, primarily from ogonori (Gracilaria) and "tengusa" (Gelidiaceae).
 W
WGel electrophoresis is a method for separation and analysis of macromolecules and their fragments, based on their size and charge. It is used in clinical chemistry to separate proteins by charge or size and in biochemistry and molecular biology to separate a mixed population of DNA and RNA fragments by length, to estimate the size of DNA and RNA fragments or to separate proteins by charge.
 W
WGelatin or gelatine is a translucent, colorless, flavorless food ingredient, commonly derived from collagen taken from animal body parts. It is brittle when dry and rubbery when moist. It may also be referred to as hydrolyzed collagen, collagen hydrolysate, gelatine hydrolysate, hydrolyzed gelatine, and collagen peptides after it has undergone hydrolysis. It is commonly used as a gelling agent in food, beverages, medications, drug and vitamin capsules, photographic films and papers, and cosmetics.
 W
WGelation is the formation of a gel from a system with polymers. Branched polymers can form links between the chains, which lead to progressively larger polymers. As the linking continues, larger branched polymers are obtained and at a certain extent of the reaction links between the polymer result in the formation of a single macroscopic molecule. At that point in the reaction, which is defined as gel point, the system loses fluidity and viscosity becomes very large. The onset of gelation, or gel point, is accompanied by a sudden increase in viscosity. This "infinite" sized polymer is called the gel or network, which does not dissolve in the solvent, but can swell in it.
 W
WA hydrogel is a crosslinked hydrophilic polymer that does not dissolve in water. They are highly absorbent yet maintain well defined structures. These properties underpin several applications, especially in the biomedical area. Many hydrogels are synthetic, but some are derived from nature. The term 'hydrogel' was coined in 1894.
 W
WLow molecular-mass organic gelators (LMOGs) are the monomeric sub-unit which form self-assembled fibrillar networks (SAFINs) that entrap solvent between the strands. SAFINs arise from the formation of strong non-covalent interactions between LMOG monomeric sub-units. As SAFINs are forming, the long fibers become intertwined and trap solvent molecules. Once solvent molecules are entrapped within the network, they are immobilized by surface tension effects. The stability of a gel is dependent on the equilibrium between the assembled network and the dissolved gelators. One characteristic of an LMOG, that demonstrates its stability, is its ability to contain an organic solvent at the boiling point of that solvent due to extensive solvent-fibrillar interactions. Gels self-assemble through non-covalent interactions such as π-stacking, hydrogen-bonding, or Van der Waals interactions to form volume-filling 3D networks. Self-assembly is key to gel formation and dependent upon reversible bond formation. The propensity of a low molecular weight molecule to form LMOGs is classified by its Minimum Gelation Concentration (MGC). The MGC is the lowest possible gelator concentration needed to form a stable gel. A lower MGC is desired to minimize the amount of gelator material needed to form gels. Super gelators have a MGC of less than 1 wt%.
 W
WNanofoams are a class of nanostructured, porous materials (foams) containing a significant population of pores with diameters less than 100 nm. Aerogels are one example of nanofoam.
 W
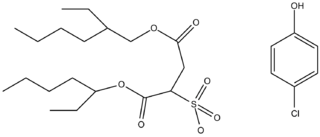
WAn organogel is a class of gel composed of a liquid organic phase within a three-dimensional, cross-linked network. Organogel networks can form in two ways. The first is classic gel network formation via polymerization. This mechanism converts a precursor solution of monomers with various reactive sites into polymeric chains that grow into a single covalently-linked network. At a critical concentration, the polymeric network becomes large enough so that on the macroscopic scale, the solution starts to exhibit gel-like physical properties: an extensive continuous solid network, no steady-state flow, and solid-like rheological properties. However, organogels that are “low molecular weight gelators” can also be designed to form gels via self-assembly. Secondary forces, such as van der Waals or hydrogen bonding, cause monomers to cluster into a non-covalently bonded network that retains organic solvent, and as the network grows, it exhibits gel-like physical properties. Both gelation mechanisms lead to gels characterized as organogels.
 W
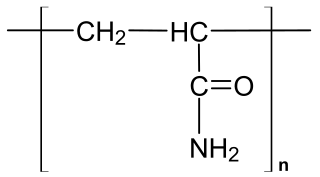
WPolyacrylamide (abbreviated as PAM) is a polymer with the formula (-CH2CHCONH2-). It has a linear-chain structure. PAM is highly water-absorbent, forming a soft gel when hydrated. In 2008, an estimated 750,000,000 kg were produced, mainly for water treatment and the paper and mineral industries.
 W
WSilica gel is an amorphous and porous form of silicon dioxide (silica), consisting of an irregular tridimensional framework of alternating silicon and oxygen atoms with nanometer-scale voids and pores. The voids may contain water or some other liquids, or may be filled by gas or vacuum. In the last case, the material is properly called silica xerogel.
 W
WA thickening agent or thickener is a substance which can increase the viscosity of a liquid without substantially changing its other properties. Edible thickeners are commonly used to thicken sauces, soups, and puddings without altering their taste; thickeners are also used in paints, inks, explosives, and cosmetics.
 W
WWater crystal gel or water beads or gel beads is any gel which absorbs and contains a large amount of water. Water gel is usually in spherical form and composed of a water-absorbing superabsorbent polymer such as a polyacrylamide.